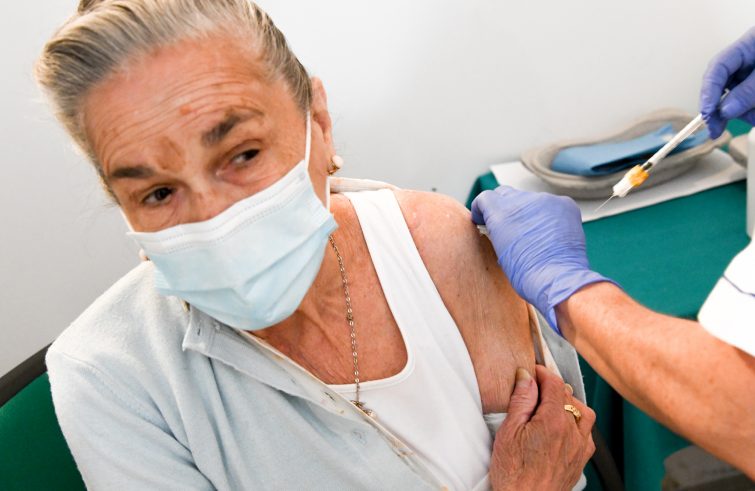
Green light for immunocompromised people, the elderly and healthcare workers. Covid-19 vaccine booster shots to kick off in Italy before long, following the approval from Health Minister Roberto Speranza on the sidelines of the G20 Health summit, confirmed by the extraordinary commissioner for Covid-19 emergency, General Francesco Francesco Figliuolo: “We plan to start administering a booster shot, which will initially affect an audience of about 3 million ‘immunocompromised persons’, i.e. people with weakened immune systems, by the end of the month. The Scientific and Technical Committee recommended a further administration of the vaccine for this population bracket back in July.” While the Italian Medicines Agengy’s Technical and Scientific Committee (Commissione Tecnico Scientifica – CTS) is holding a meeting to decide on the third dose of the vaccine, SIR contacted by phone Patrizia Laurenti, Head of the Hospital Hygiene Unit and Director of the Vaccination Centre at Agostino Gemelli Hospital, Rome.
Professor, how important is it to begin rolling out the third dose as soon as possible?
The priority is to
give a booster shot to immunocompromised people, those who, either on account of their medical condition or old age, have a weaker antibody response than people with normal immune systems.
 That will be the priority in the coming months of 2021. We must not forget, however, that a booster dose is required for healthcare workers, almost all of whom were vaccinated between January and February. To this end, the healthcare facilities where they work must start planning starting today.
That will be the priority in the coming months of 2021. We must not forget, however, that a booster dose is required for healthcare workers, almost all of whom were vaccinated between January and February. To this end, the healthcare facilities where they work must start planning starting today.
Meanwhile, we must not forget the flu shot, which the Ministry of Health plans to start administering in early October. In short, that means the two immunisation programmes are back on track.
Will mRna vaccines – Pfizer and Moderna – also be given to persons who completed the first cycle with Astrazeneca or Johnson&Johnson?
Presumably so. Heterologous vaccination has been scientifically validated and many studies have shown that mixing vaccines can elicit a stronger/longer-lasting response. Furthermore, mRNA vaccine production has continued and we have it in large supplies.
In addition to the ‘die-hard’ anti-vaxxers, the vaccine-hesitant group includes a large number of people who, in addition to not protecting themselves, are slowing down the attainment of herd immunity in the belief that the Covid vaccine is an experimental drug.
This is a mistaken belief in terms of the definition of ‘experimental drug’. The Covid vaccine has successfully completed all stages of clinical trials. Its pace was accelerated by a combination of events: the urgency dictated by the pandemic, the availability of funds, the pooling of scientists from across the globe. The trial phase is followed by so-called post-marketing surveillance, which occurs after the marketing of the product for the population at large. Billions of doses have been administered in the past eight months, and surveillance data on adverse events are extremely reassuring. That’s why
to continue speaking of an ‘experimental drug’ is a conceptual error.
Which procedures will have to be followed? Does it require authorisation from the Italian Medicines Agency?
In this case it is not mandatory. Available evidence-based data entitles EU Member States to decide independently without waiting for specific authorisation.
The idea of introducing compulsory vaccination is gaining ground…
At this point I think it is necessary to reach out to those three million people over 50 who have not been vaccinated for unspecified reasons unknown to us. Nonetheless, a frank debate with them is recommended. We are available:
the “vaccine-hesitancy” issue is important; these people must be listened to and motivated.












